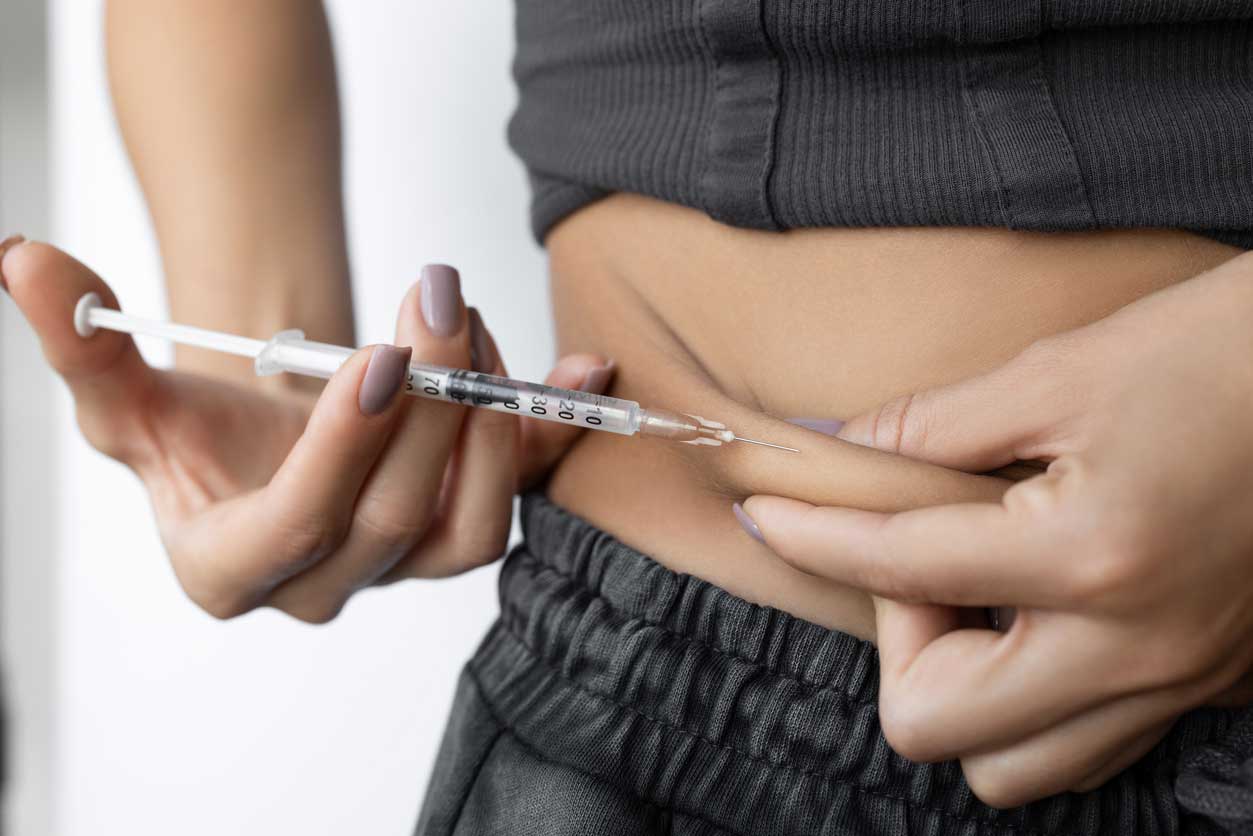
Adipose tissue was identified as being a great source for stem cells. While harvesting and isolating stem cells from adipose tissue takes considerably longer than other methods, people can choose to store them and use them at a later date. Another important advantage of fat derived stem cell injections is that they are harvested from the same patient and do not involve the use of embryos.
Call to Schedule Free Consultation at Over 80 Centers Worldwide!
- Home
- Success Stories
- About
- Stem Cells
- Conditions
Autoimmune
Cardiovascular
Endocrine
Gastrointestinal
Liver
Musculoskeletal
Neurological
Other
Respiratory/Pulmonary
Reproductive
Renal/Kidney
Urologic
- Celebrities
- Locations
- Home
- Success Stories
- About
- Stem Cells
- Conditions
Autoimmune
Cardiovascular
Endocrine
Gastrointestinal
Liver
Musculoskeletal
Neurological
Other
Respiratory/Pulmonary
Reproductive
Renal/Kidney
Urologic
- Celebrities
- Locations