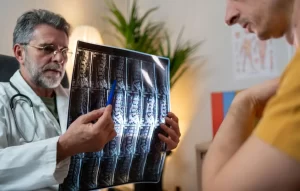
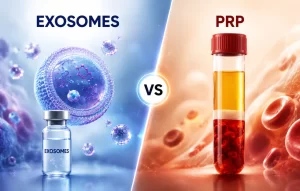
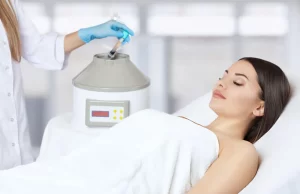
Why Athletes Quit — And Why They Don’t Have To Most athletes don’t stop competing because they stop caring. They stop because their bodies won’t cooperate. Chronic joint pain, stiffness, and sluggish recovery are the real reasons careers end — not a loss of passion....
Read MoreCall to Schedule Free Consultation at Over 80 Centers Worldwide!
- Home
- Success Stories
- About
- Stem Cells
- Conditions
Autoimmune
Cardiovascular
Endocrine
Gastrointestinal
Liver
Musculoskeletal
Neurological
Other
Respiratory/Pulmonary
Reproductive
Renal/Kidney
Urologic
- Celebrities
- Locations
- Home
- Success Stories
- About
- Stem Cells
- Conditions
Autoimmune
Cardiovascular
Endocrine
Gastrointestinal
Liver
Musculoskeletal
Neurological
Other
Respiratory/Pulmonary
Reproductive
Renal/Kidney
Urologic
- Celebrities
- Locations